(English follows Japanese)
採取する場所で性質が変わる炭素系ナノ粒子層
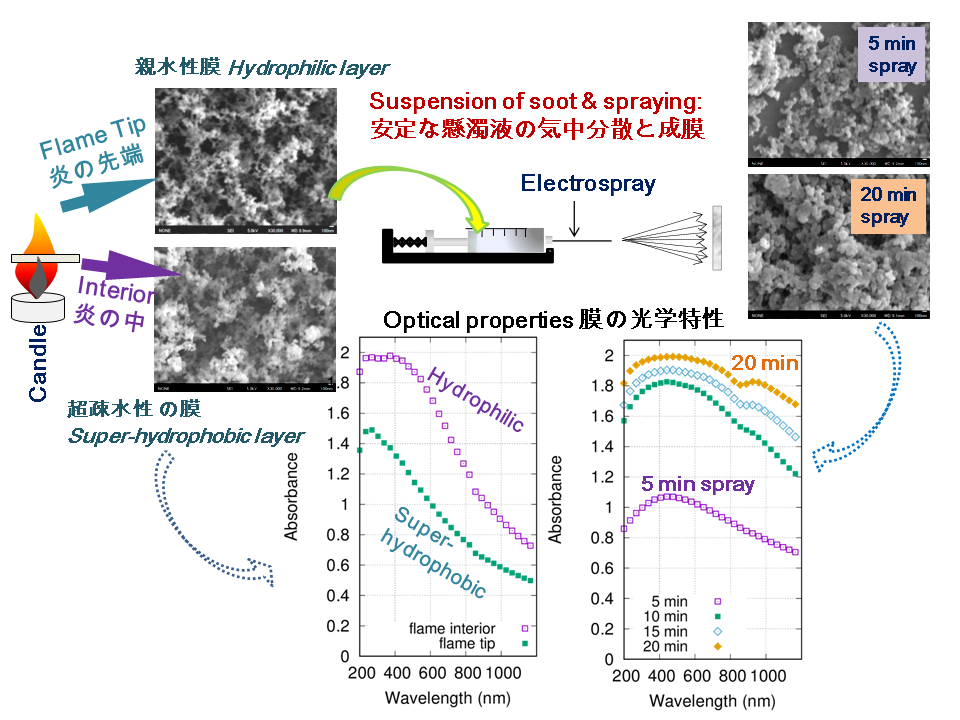
同じろうそくの炎から出る「すす」でも、集める場所によって、水を強くはじく材料にも、水になじむ材料にもなる――。この研究では、ろうそく燃焼で生じる炭素系すす粒子を使い、その粒子層の表面性質や構造、光吸収特性を調べました。
研究では、2つの作製ルートが用いられました。1つ目は、ガラス基板をろうそくの炎の近くに置き、すす粒子を直接堆積させる方法です。2つ目は、いったん集めたすすをエタノール中に分散させ、静電噴霧法(エレクトロスプレー)で再び基板上に堆積させる方法です。
最も印象的な結果は、すすを集める位置によって水との相性が大きく変わったことです。炎の内部から集めたすす粒子層では、水滴が丸く保たれ、接触角は約151°となり、超撥水性を示しました。一方、炎の先端で集めたすす粒子層では、水滴が吸い込まれ、接触角は約5°となり、親水性を示しました。つまり、同じろうそく由来のすすでも、採取する位置を変えるだけで、まるで「水をはじく黒い膜」と「水になじむ黒い膜」のように異なる性質を示したのです。
ラマン分光分析では、炎の内部と先端で得られた粒子層の D バンドと G バンドの強度比が異なることが示され、炭素構造の違いが表面性質に関係している可能性が示されました。また、エレクトロスプレーによる再堆積では、すす粒子の主要な結合構造は大きく変化しないことも確認されました。
さらに、光吸収測定では、すす粒子層が紫外から近赤外領域まで吸収特性を示し、作製方法や粒子層の形態によってスペクトルの傾きが変化することが示されました。
この研究は、身近なろうそくのすすが、採取条件や堆積方法によって異なる機能を示す炭素ナノ粒子材料になり得ることを示しています。表面の濡れ性や光吸収を制御できる粒子層を、より扱いやすく作製するための基礎的な知見です。

Using “soot” from a candle burning, we discovered /developed two assembly routes to form hydrophilic or hydrophobic layers of carbonaceous nanoparticles.
ロウソク燃焼の「煤」を用いて、親水性または疎水性を有するナノ粒子膜の作製する方法を開発。5000年前に中国で開発された、従来の「墨」技術と同じかどうか?

Open access (Full PDF): https://doi.org/10.4209/aaqr.2017.10.0426
Carbon-based nanoparticle layers with tunable surface properties**
Can the same candle soot become either water-repellent or water-friendly? This study showed that carbonaceous soot particles from candle combustion can form particle layers with very different surface properties depending on where the soot is collected from the flame.
Two preparation routes were examined. In Route 1, soot aerosol particles were deposited directly onto a substrate placed near the candle flame. In Route 2, collected soot particles were dispersed into a suspension and then redeposited onto a substrate by electrospray.
The key finding was the strong contrast in wettability. Soot collected from the flame interior formed a superhydrophobic layer with a water contact angle of 151.0 ± 1.3°, while soot collected from the flame tip produced a hydrophilic layer with a contact angle of 5.2 ± 0.9°. In simple terms, changing the collection position turned candle soot into either a “water-repelling black layer” or a “water-absorbing black layer.”
Raman spectroscopy showed different D- and G-band intensity ratios between soot layers from different flame regions, suggesting that carbon bonding structures may contribute to their wettability. The study also found that electrospray redeposition did not significantly alter the main bond structure of soot particles, while offering a more controllable way to form particle layers.
Optical measurements further showed absorption features from ultraviolet to near-infrared wavelengths, with spectral changes depending on the preparation route and morphology of the soot layer.
Overall, this work demonstrates that candle-derived soot is not just a combustion byproduct but a tunable carbonaceous nanoparticle material. It provides basic insight into controlling wettability and optical properties through simple deposition conditions.

